Cognitive peptides: promise, evidence, and a retraction
Research-first walkthrough of cognitive peptides, what Semax, Selank, and DSIP actually do, which claims don't hold up, and what to watch in 2026.
A foundational paper in cognitive-peptide research was retracted in April 2025. The peptide it described, Dihexa, did not disappear from research websites, Reddit threads, or supplement stacks. That gap, between what the public chatter says about cognitive peptides and what the data actually supports, is the honest starting point for anyone considering this class.
Cognitive peptides work by modulating BDNF and synaptic plasticity, not by adding a new neurotransmitter#
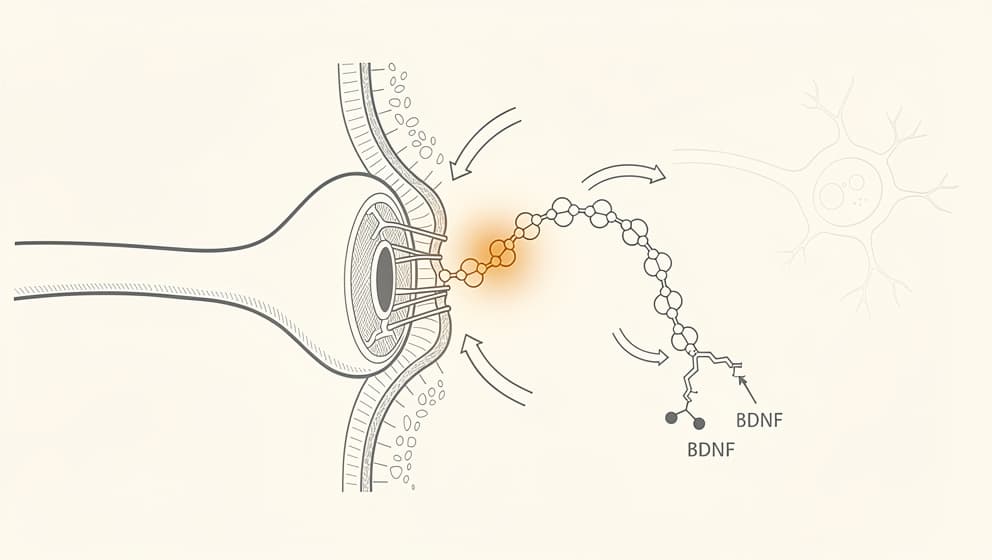
The class-defining mechanism of cognitive peptides, often called nootropic peptides in consumer contexts, is neurotrophic signaling rather than direct neurotransmission. Most non-peptide nootropics (caffeine, L-theanine, modafinil, the racetam family) act on neurotransmitter receptors: adenosine, GABA, dopamine, or acetylcholine. The effect shows up within an hour and fades with the half-life. Cognitive peptides operate on a slower, structural timescale. They change the expression of proteins that build and strengthen synapses.
Brain-derived neurotrophic factor (BDNF) is the load-bearing example. BDNF is a protein that supports neuron survival, promotes synapse formation, and strengthens the connections between neurons when learning occurs. It is the molecular route through which experience physically changes the brain. Studies have shown higher BDNF expression is associated with faster memory formation, better recall, and resilience against stress-induced depression in both animal and human work (Asua et al., 2018, PMID 29030286). Exercise raises BDNF. So do selective serotonin reuptake inhibitors, though more slowly. So do several peptides.
Semax is the cleanest example of a peptide with a direct BDNF effect. A single 50 µg/kg intranasal dose in rats produced a 1.4-fold increase in hippocampal BDNF protein and a roughly 3-fold increase in BDNF messenger RNA within thirty minutes (Dolotov et al., 2006). The increase was not subtle or transient. BDNF's downstream receptor TrkB also rose by about 1.6-fold. In the same animals, learning performance on a standard conditioned-avoidance test improved. The mechanism chain, peptide to BDNF to TrkB to synaptic plasticity to measurable behavioural change, is short, testable, and has been replicated.
Selank takes a different path to a related endpoint. Research has shown its primary effect appears to be on GABA-receptor subunit expression and monoamine balance, particularly serotonin and dopamine. Clinical studies in Russia show anxiolytic effects comparable to benzodiazepines without the sedation, cognitive dulling, or dependence profile typical of that drug class (Zozulya et al., 2008). Selank also appears to upregulate BDNF secondarily. The reduction in anxiety, in practice, translates into cleaner working memory and attention on the days Selank is dosed.
The important conceptual difference between nootropic peptides and traditional small-molecule nootropics is this: peptides ask the brain to build (the neuroplasticity argument). Nootropics ask the brain to work harder with what it already has. That distinction shapes dose cadence (daily for weeks versus ad-hoc), the expected timeline to effect (a month or more rather than an hour), and the interpretation of subjective feedback. A peptide that "does nothing on day three" has not failed. A nootropic that does nothing in forty minutes has.
Four cognitive peptides have meaningful human data, and none are FDA-approved in the United States#
The cognitive-peptide conversation is dominated by four compounds with enough clinical signal to discuss seriously. Each belongs to a different mechanism family.

| Peptide | Origin | Typical research dose | Route | Evidence tier | Regulatory status (April 2026) |
|---|---|---|---|---|---|
| Semax | Russia, 1990s | 100–300 µg | Intranasal / subcutaneous | Moderate clinical (Russian trials) | Approved in Russia; not FDA or EMA |
| Selank | Russia, 2009 | 100–300 µg | Intranasal / subcutaneous | Moderate clinical (Russian trials) | Approved in Russia; not FDA or EMA |
| DSIP | Switzerland, 1970s | 100–300 µg | Subcutaneous, pre-bed | Preclinical + small open-label | Not approved anywhere |
| Cerebrolysin | Austria | Physician-administered | IV or IM, 4-week courses | Strong RCT base (772-patient meta-analysis) | Registered in ~40 countries; not US, not approved in Norway |
Semax. A 7-amino-acid analog of ACTH(4-10) with no hormonal activity. Developed at the Institute of Molecular Genetics of the Russian Academy of Sciences and registered in Russia in the 1990s. Primary indications (in Russian practice): stroke recovery, cognitive dysfunction, and attention disorders. Route: intranasal or subcutaneous. Typical research dose range: 100 to 300 mcg per administration, once or twice daily. The BDNF-TrkB upregulation described above is its best-documented mechanism. Human stroke-recovery data from Russian centres shows accelerated functional recovery versus standard care, though the trials are small and published predominantly in Russian journals.
Selank. A 7-amino-acid synthetic analog of the immune peptide tuftsin. Same institute of origin. Approved in Russia in 2009 for generalized anxiety disorder and neurasthenia. Route: intranasal or subcutaneous. Research dose range: 100 to 300 mcg per administration. The 2008 Russian trial compared 30 Selank patients against 32 medazepam patients on the Hamilton Anxiety scale. Anxiolytic effect was similar between arms, but the Selank group retained antiasthenic and mild psychostimulant effects (Zozulya et al., 2008). Research suggests Selank acts as a positive allosteric modulator at GABA-A sites, a mechanism distinct from how benzodiazepines bind the same receptor, which may be the reason for the cleaner tolerability profile.
DSIP (Delta Sleep-Inducing Peptide). A 9-amino-acid nonapeptide originally isolated from rabbit cerebral venous blood during electrical sleep induction in the 1970s. DSIP's evidence base is older and thinner than Semax or Selank. Animal work supports a modest effect on delta-wave sleep promotion and stress-axis dampening. Human data is limited to small open-label series. Route: subcutaneous, 1 to 3 hours before bedtime. Research dose range: 100 to 300 mcg. DSIP sits in the cognitive cluster not because it improves cognition directly but because sleep architecture is upstream of almost every cognitive outcome worth measuring.
Cerebrolysin. An outlier in this list. Not a single peptide but a standardised mixture of low-molecular-weight peptides and amino acids derived from porcine brain tissue. Registered across roughly 40 countries in Europe, Asia, and Latin America. Administered intravenously or intramuscularly in clinical settings, typically in 4-week courses. Cerebrolysin has the strongest RCT base of any compound in this category: a 2015 meta-analysis pooled six randomized placebo-controlled trials with 772 patients and found a statistically significant benefit in mild-to-moderate Alzheimer's disease. Response rate was 66.9% with Cerebrolysin versus 41.8% with placebo (Gauthier et al., 2015). The effect size at four weeks on cognitive function was a standardised mean difference of minus 0.40 (95% CI minus 0.66 to minus 0.13).
Notable absences from this list: Noopept (technically not a peptide; it's a small-molecule prodrug of a peptide fragment), Pinealon (mechanism claims exist but human data is sparse), and the broader family of "brain peptides" marketed in wellness clinics that often lack specific active compound identification. Research quality varies by orders of magnitude across this space. A proper name and a PubMed ID are the minimum filter. Readers specifically weighing Semax against Selank as a first cognitive peptide protocol can go deeper in the Semax vs Selank comparison.
Dihexa and a handful of others earned reputations on research that was later retracted#
This is the uncomfortable part of the cognitive-peptide story, and it deserves its own section. The cognitive-peptide field carries a handful of compounds whose public reputations outpace the published evidence, and Dihexa is the clearest case study.
Dihexa is a small synthetic peptide derived from angiotensin IV. It became famous in biohacker circles through a single comparative claim, that it was reportedly "seven times more potent than BDNF" at promoting synaptogenesis, which traced back to one foundational paper: Benoist et al., 2014, published in the Journal of Pharmacology and Experimental Therapeutics. That paper established the proposed mechanism, Dihexa binding with high affinity to hepatocyte growth factor (HGF) and activating the c-Met receptor system, a pathway that supports new synaptic connections and dendritic spine growth.
In 2021, Retraction Watch reported that multiple papers from the originating laboratory (the Harding-Wright lab at Washington State University) had received expressions of concern. The investigation centred on image manipulation by Leen Kawas, a former graduate student from the lab who went on to become chief executive of Athira Pharma, a biotech company commercialising Dihexa derivatives. Benoist et al. 2014 was formally retracted in April 2025. The retraction notice cites figure manipulation that cannot be remediated by the original authors.
What does that mean for the compound?
It means the headline claim, the specific HGF/c-Met mechanism, cannot be cited from that paper going forward. Independent replication of the mechanism remains incomplete as of this writing. The broader class of angiotensin IV-derived peptides has other supporting literature, and Dihexa may still have cognitive effects for all the ordinary pharmacological reasons. But the public record has shifted. Consumers and researchers should treat Dihexa's mechanism as an open question, not a settled fact.
The wider point matters beyond Dihexa. The cognitive-peptide field suffers from a long tail of compounds whose reputations rest on a single study, often from a single laboratory, often unpublished in a major peer-reviewed venue. P21 is another example, described in a small number of rodent studies with neurogenic effects, with no human evidence and no independent replication in the consumer-accessible timeline. When a class of compounds produces subjective effects that are hard to measure in a short trial (a common feature of anything working on BDNF or synaptic plasticity), and when that class sits in a regulatory grey zone outside conventional pharmaceutical development, the normal error-correction mechanisms of science work more slowly than they do for, say, a cholesterol drug. A retraction at the foundational layer in a small field has an outsized effect.
The honest default for compounds in this category is skepticism calibrated to the evidence: how many human trials, what sample sizes, how many labs, how old is the evidence, has anyone tried to replicate it, has anything been retracted. Compounds that pass this filter make it into Klarovel's catalogue. Compounds that do not pass do not.
Delivery matters: intranasal, subcutaneous, and oral each change the peptide that reaches the brain#
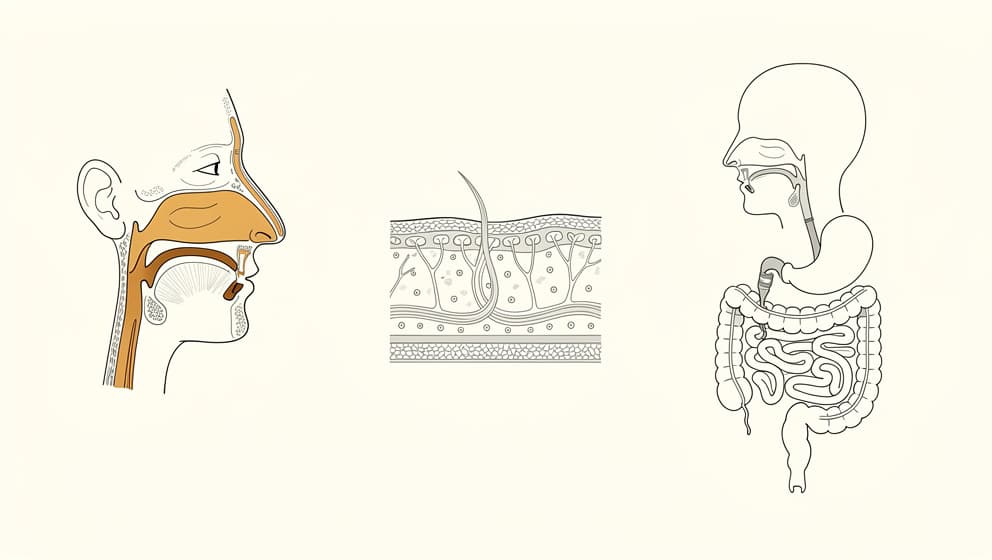
Route of administration is one of the most misunderstood variables in cognitive-peptide use. Two cognitive peptides with identical molecular structure can behave differently depending on how they reach the brain. The same peptide dosed three different ways is effectively three different interventions.
Intranasal. Small peptides absorbed through the olfactory and respiratory epithelium can bypass the blood-brain barrier and reach cerebrospinal fluid directly. For Semax and Selank, this is the route Russian clinical practice relies on, and it is the route most of the mechanism literature uses. Intranasal absorption is not 100% efficient; a meaningful fraction of the dose is swallowed or absorbed systemically. But the fraction that does reach the olfactory bulb is in the central nervous system within minutes. That is what makes the subjective onset of intranasal Semax notably faster than the same dose delivered subcutaneously.
Subcutaneous. Small peptides delivered subcutaneously enter systemic circulation through capillary absorption and reach the brain to the extent that they cross the blood-brain barrier. Semax, Selank, and DSIP all have sufficiently small molecular sizes and favourable physicochemical properties to cross at therapeutic levels when dosed correctly. The onset is slower than intranasal, the subjective effect is typically smoother, and the dose can be more precisely controlled. Klarovel's peptide calculators handle the mcg-to-insulin-unit conversion for common formulations (peptide reconstitution calculator).
Oral. The hardest route. Most peptides are hydrolysed in the stomach, digested in the small intestine, or cleared by first-pass hepatic metabolism before reaching systemic circulation. Oral bioavailability for unmodified peptides in this class is typically below 1%. Claims that oral peptide supplements produce measurable cognitive effects are thus mechanistically implausible without either (a) structural modification to resist digestion, or (b) delivery technology (enteric coating, absorption enhancers) that the product does not advertise. A small number of oral peptide drugs have been approved (oral semaglutide, for example, uses a permeation enhancer) but these are pharmaceutical-grade formulations, not supplement-aisle preparations.
Intravenous / intramuscular. Reserved in practice for Cerebrolysin, which is typically administered as a 4-week course of IV or IM injections in a clinical setting. This is not a route for self-administered cognitive optimisation. It is a route for patients under physician supervision.
The practical upshot: when comparing two peptide protocols that look similar on paper, check the route first. An "intranasal Semax microdose" and a "subcutaneous Semax 300 mcg" protocol are different experiments producing different biology.
Regulation is uneven: Russia-approved, EU-unapproved, and a US reclassification worth watching#
Cognitive peptides sit in a regulatory landscape that looks different depending on which country's stamp a reader values most. The same compound can be a registered medicine in one country, a bulk research substance in another, and effectively unavailable in a third.
Russia. Semax has been licensed since the mid-1990s for stroke recovery and cognitive dysfunction. Selank has been licensed since 2009 for generalized anxiety disorder and neurasthenia. Both are on the Russian essential medicines list. The clinical experience base is real but is mostly published in Russian-language journals, which limits international peer review and replication.
European Union and Norway. Neither Semax nor Selank is approved as a medicinal product in the EU or Norway. Legemiddelverket, Norway's Medicines Agency, classifies unapproved peptides as medicinal products that cannot be legally imported for personal use without a prescription obtained in accordance with the Medicines Act (Legemiddelloven). Cerebrolysin is marketed in several European countries, though not in Norway under that name. DSIP has no approved medicinal product status in the EU.
United States. The FDA has not approved any of the peptides discussed here for any cognitive indication. In September 2023, the FDA placed several peptides on a list of bulk drug substances presenting significant safety risks, restricting their use in compounding pharmacies. In early 2026, reporting suggested reclassification discussions were underway that may change the rules for certain peptides under compounding pharmacy preparation. The details remain in flux and warrant ongoing attention, not premature conclusions.
The regulatory patchwork has two practical implications. First, sourcing quality varies enormously. A peptide ordered from a research-chemistry supplier is not quality-controlled the way a pharmacy-compounded product is. Independent third-party assays regularly find large variations in peptide content, purity, and endotoxin levels. Second, the legal framing differs by jurisdiction. Klarovel operates in Norway, where compliance with Legemiddelloven and the associated import rules is the binding constraint, and where partnerships with licensed providers are the route to access, not parcel-forwarding workarounds.
The first eight weeks of a cognitive peptide protocol rarely feel like a switch flip#
The most common mistake cognitive peptides make on their users is an evaluation mistake, not a biological one. Readers come to this class expecting something adjacent to a stimulant or an antidepressant: noticeable on-switch, discernable off-switch, subjective feedback that confirms or denies the compound is doing what the label promises. Cognitive peptides do not behave that way. Neither do the mechanisms they target. BDNF upregulation is a structural change in how synapses strengthen, not a minute-to-minute performance lever. Protocol evaluation needs to match the biology.
A reasonable subjective timeline for a cognitive peptide like Semax or Selank, dosed at the research ranges described earlier, looks something like this. Weeks one and two are often unremarkable. Some users report a subtle mood-floor lift or less anxiety scaffolding by day ten, but many notice nothing. That is expected. The peptide is at steady state in circulation within a week of daily dosing. The downstream changes it triggers take longer to translate into noticeable cognition.
Weeks three and four are where most people form an opinion. By this point, a genuine signal, if there is one, tends to show up in the granular measures rather than the headline ones. Sleep architecture often changes first: fewer 3am wakeups, less next-morning fog. Task-switching cost on demanding work drops. For Selank users the social-anxiety floor shifts, which is often the first clear signal. What usually does not change, and is not supposed to, is the peak of a peak day. Cognitive peptides raise the floor. They do not raise the ceiling.
Weeks five through eight is where consolidation happens. The protocol is either clearly helping on the measures that were worth tracking at baseline, or it is not. The honest next step in either case is to stop and reassess. A cognitive peptide that is doing its job justifies a cycle off, a bloodwork check, and a decision about whether to continue into a second block. A cognitive peptide that is not doing its job does not earn more runway; the signal after eight weeks of consistent dosing is most of the signal there is.
Three practical rules make this easier to run. First, define the measures at baseline, before the first dose, in writing. Sleep hours, a one-to-ten morning clarity score, and a one-line description of the specific cognitive friction the protocol is supposed to address. Without baselines, every week is noise. Second, dose at a consistent time of day. Mid-morning for Semax. Mid-day or pre-stressor for Selank. Pre-bed for DSIP. Circadian alignment matters with neurotrophic compounds in a way it does not with stimulants. Third, read the caution signals seriously. A persistent headache after day seven, a blood-pressure rise above baseline, or injection-site inflammation that does not settle within 24 hours is the protocol telling the user something. The correct response is usually to pause and reassess, not to push through.
Frequently asked questions about cognitive peptides#
Are cognitive peptides FDA-approved? None of the peptides discussed here (Semax, Selank, DSIP, Cerebrolysin) are FDA-approved for any cognitive indication in the United States. Semax and Selank are approved as medicines in Russia. Cerebrolysin is registered in roughly 40 countries across Europe, Asia, and Latin America, but not the US. In Norway, none of these peptides hold a marketing authorisation from Legemiddelverket; they are classified as unapproved medicinal products.
How long does it take to notice whether Semax or Selank is working? Most users see no clear signal in weeks one and two. Weeks three and four are where a genuine effect tends to show up in granular measures (sleep quality, task-switching cost, social-anxiety floor) rather than headline peak-performance. The honest evaluation point is eight weeks of consistent dosing at the research dose range.
Do any peptides help with ADHD? Semax has the most mechanistic relevance here because of its BDNF-TrkB effect, and Russian practice uses it for attention disorders. Human data for adult ADHD specifically is limited to small studies and case series. Selank's anxiolytic effect can reduce anxiety-driven executive dysfunction, which is sometimes mistaken for ADHD. Neither compound is FDA-approved for ADHD, and neither substitutes for an assessment and evidence-based treatment through a clinician.
Is Semax legal in Norway? Semax is not an approved medicine in Norway. Legemiddelverket classifies unapproved peptides as medicinal products that cannot legally be imported for personal use without a prescription obtained in accordance with the Medicines Act. Purchasing from unlicensed research-chemistry suppliers carries both legal and quality-control risks.
Can cognitive peptides be stacked? Semax and Selank are sometimes used together in Russian protocols, with Semax's focus-enhancing effect balanced by Selank's anxiolytic profile. DSIP is orthogonal to both and targets sleep architecture rather than daytime cognition. Cerebrolysin is rarely stacked outside clinical settings because it is administered by a clinician in 4-week IV or IM courses. Klarovel's engine handles stacking rules automatically based on questionnaire data, medication review, and biomarker status.
Read more on Klarovel#
- Selank Peptide: Russian-Approved, Not FDA-Authorized: Selank peptide is a Russian-approved anxiolytic that modulates GABA-A and enkephalins.
- Semax Peptide: Approved in Russia, Unscheduled in the US: Semax peptide is a Russian-approved nootropic that raises BDNF and shows neuroprotective effects after stroke.
Klarovel's position: the engine decides which cognitive peptide (if any) fits your profile#
Cognitive-peptide selection is not a generic recommendation. A cognitive peptide that works beautifully for a thirty-five-year-old with post-COVID brain fog is not necessarily the right cognitive peptide for a sixty-year-old with early mild cognitive impairment, nor for a twenty-eight-year-old with generalized anxiety that spills into executive function. The right answer depends on what the reader is actually optimising for: peptides for focus under heavy cognitive load, peptides for memory consolidation, peptides that quiet the anxiety that spills into brain fog, sleep architecture that is quietly degrading every other cognitive outcome, or structural neuroprotection in the context of a family history of decline. Each of these points to a different peptide (or to no peptide at all, with a different intervention upstream).
Klarovel's approach is to separate the two questions most wellness content conflates. The first question is diagnostic: what are the specific symptoms, biomarkers, and goals, and what is already in place (medications, existing conditions, bloodwork)? The second question is therapeutic: given the diagnostic picture, what protocol has the best evidence-to-risk ratio? Klarovel's protocol engine handles the therapeutic question by running a rules-based pipeline over a catalogued set of peptides, including contraindication screening, biomarker-aware dose modulation, and stacking rules. The diagnostic question is captured upstream, in the health questionnaire, which also surfaces concerns that may mean no peptide is the right answer for the moment.
The result is a protocol that is specific to the individual rather than a list of peptides pulled from a blog post. It also means the cognitive-peptide conversation becomes one thread inside a broader conversation about energy, stress, sleep, and metabolism, which is usually where cognitive outcomes actually come from. A well-designed protocol for someone asking about peptides for focus may actually start with correcting ferritin, vitamin D, or thyroid status, and return to Semax or Selank only if the symptoms persist after the foundations are addressed. The same logic applies to peptides for memory: structural cognitive interventions only pay back when the metabolic and inflammatory background is reasonable.
For readers interested in the science-first path into this category, start with the questionnaire rather than a product page. The questionnaire is free, takes about twelve minutes, and produces a personalized evaluation that factors in both what the research suggests and what your individual physiology appears to need. The cognitive peptides conversation in 2026 is clearer than it was five years ago. The best use of that clarity is to skip the hype cycle and get a protocol that reflects your specific situation. Complete the Klarovel health questionnaire to see what the engine recommends, or browse the peptide catalogue to understand what is available under Klarovel partnerships.
Keep reading
Selank Peptide: Russian-Approved, Not FDA-Authorized
Selank peptide is a Russian-approved anxiolytic that modulates GABA-A and enkephalins. The mechanism, the 62-patient trial, and 2026 regulatory reality.
Semax Peptide: Approved in Russia, Unscheduled in the US
Semax peptide is a Russian-approved nootropic that raises BDNF and shows neuroprotective effects after stroke. The science and the 2026 regulatory reality.
BPC-157: mechanism, evidence, and what the research actually shows
BPC-157 is the most-searched healing peptide. Here's the mechanism, the preclinical evidence, the limited human data, and how to think about the protocol.